
Recently, Yang Sheng, the director of the National Medical Products Administration, stated, "The number of innovative drugs under research in China has accounted for 30% of the global total, and the scale of China's pharmaceutical industry ranks second in the world." In this wave of innovation, cell and gene therapy (CGT), as a highly anticipated "star track", is accelerating at an unprecedented speed.

Looking back over the past four years, China's CGT has rapidly moved from the "approval of the first product" to the "period of industrial acceleration". Up to now, nine CGT drugs have been officially launched on the market, covering three core technologies: CAR-T, stem cells and gene therapy. They are mainly used to treat hematological malignancies, transplantation-related complications, and some hereditary rare diseases.
What is more worthy of attention is that the medical insurance and commercial insurance mechanisms are also breaking through simultaneously: from "not reimbursable" to "having a mechanism", from "full self-payment" to "diversified payment", the accessibility of high-value therapies is quietly changing. This not only means that more patients will truly benefit, but also marks that China's CGT is moving from "scientific research breakthroughs" to "system acceptance".
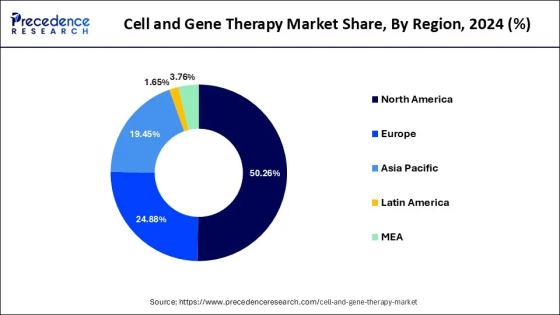
China's position in global coordinates
Looking globally, CGT is still in a period of rapid development. According to the quarterly reports released by ASGCT and Citeline, in the first quarter of 2025, new drugs for cell, gene and RNA therapies were all approved, among which CAR-T remained the main growth driver.
The United States still leads in approval speed and commercialization paths, but regulation is becoming stricter. Starting from the end of 2024, the FDA will require all CD19 and BCMA CAR-T products to add safety warnings related to "T-cell malignancies" in their instructions and strengthen long-term follow-up requirements. This indicates that global regulation is shifting from "approving quickly" to "approving steadily".
In contrast, China has caught up in terms of product quantity, review efficiency and technical paths: from having only one CAR-T product to the successive approval of stem cell therapy and AAV gene therapy, China's CGT industry is building a more complete "product spectrum".
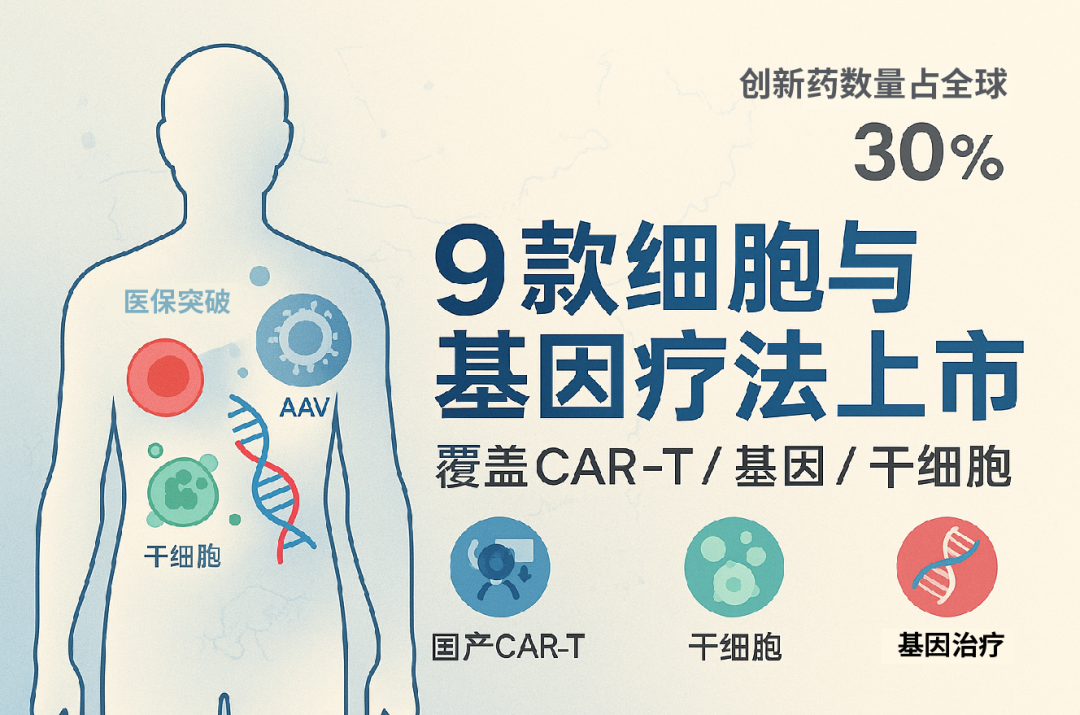
Nine cell and gene therapy drugs have been launched on the market
As of August 2025, a total of 9 CGT drugs have been approved for marketing in China, covering three major mainstream technical routes and applied in multiple high-value clinical scenarios. The following table is a list of all currently available products:
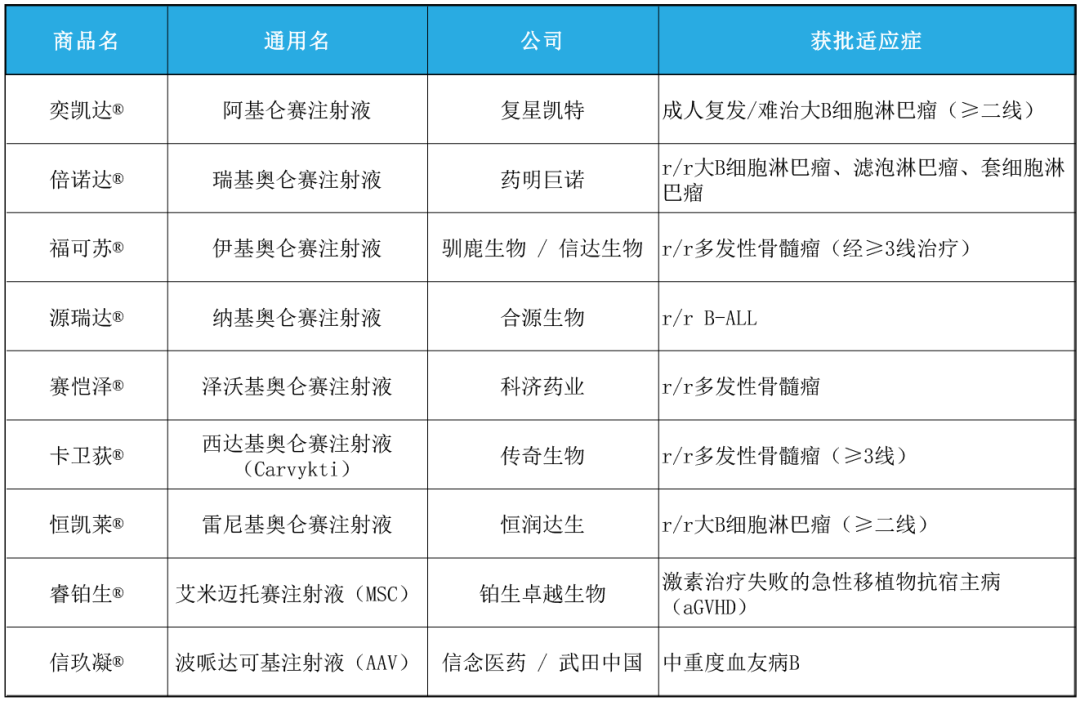
These products not only have achieved a leap from zero to one in clinical pathways, but also represent a significant improvement in China's industrial maturity in multiple key technical directions such as CAR-T, stem cells, and AAV delivery systems.
The payment mechanism is reshaping accessibility
One of the biggest problems that cell and gene therapy has faced in the past was that it was "effective but too expensive." But this situation is gradually changing.

In early 2025, the National Healthcare Security Administration released the initial review list of the first batch of innovative drugs for commercial insurance, covering 121 innovative products, including 5 CAR-T therapies. This means that high-value therapies are attempting to gradually establish a sustainable payment loop through the approach of commercial insurance taking the lead and medical insurance following up.
Meanwhile, some enterprises are also trying out new pricing strategies such as "pay based on efficacy" and "prepayment + refund". For instance, Fosun Kite has launched the "pay after reaching efficacy" policy, as well as the multi-region people's welfare insurance compensation plan in combination with the stem cell drug "Ruibosheng". These explorations have laid a practical foundation for the popularization of high-value innovative drugs.

Three points of observation for the future
Broader indications: From hematological malignancies to solid tumors and autoimmune diseases
New targets such as CLDN18.2, GPC3, and BCMA-TCR are being advanced at an accelerated rate. Clinical verification is currently underway in the directions of solid tumors/immune diseases including gastric cancer, liver cancer, and lupus erythematosus.
Production technology Innovation: From "Customization" to "Standardization"
Automated cell preparation, universal CAR-T (Allo-CAR-T), and mRNA delivery systems will become the key variables determining cost and accessibility in the next stage.
Dual-track optimization of supervision and payment
As more products are included in channels such as "priority review" and "conditional approval", the review efficiency has been further enhanced. Meanwhile, a dynamic combination mechanism of medical insurance, commercial insurance and the proportion of out-of-pocket expenses for patients is gradually taking shape.

Outlook
From "the first product to hit the market" to "nine products launched simultaneously", from "clinical breakthroughs" to "system acceptance", China's cell and gene therapy is at a critical stage of accelerated growth. This is a truly "from 0 to 1" process for China's biopharmaceutical industry. The next question is no longer "whether it exists or not", but "whether it can be popularized", "whether it is sustainable" and "whether it is useful or not". And these are precisely the new propositions that China's CGT needs to answer with facts in the next stage.
It can be foreseen that China's CGT industry will continue to thrive under the combined effect of capital, talent and the market, not only serving domestic patients but also providing "Chinese solutions" on a global scale. As the industry has commented, cell and gene therapy will become the new generation of mainstream therapies following small molecule targeted drugs and monoclonal antibodies. Under this historical opportunity, China is expected to achieve a leap from "introduction and digestion" to "innovation output". We have every reason to believe that as more therapies come onto the market and become accessible, the once "incurable diseases" will gradually be overcome, and the golden age of the CGT industry is accelerating its arrival.